A Higher Standard for Stem-Cell biologics
TAVRA Biosciences supplies cGMP-manufactured stem cell biologics used in orthopedic and regenertive medicine clinical settings.

Human Bone Marrow Mesenchymal Stem Cells
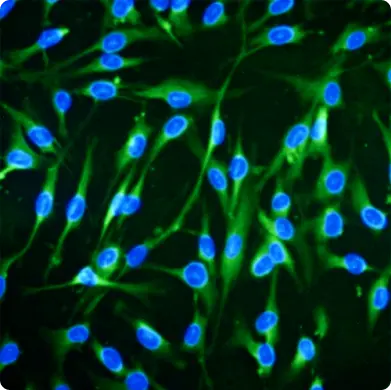
TAVRA offers bone marrow-derived mesenchymal stem cells (BM_MSCs) from young healthy donors (18-25 years old) which have demonstrated significant results alleviating pain, increasing joint function, and speeding wound healing in clinical studies.
Our product is manufactured in an FDA-registered, cGMP-compliant, ISO 7-certified cleanroom and available off the shelf, ready for immediate use at the point of care. Each lot is released with a third-party verified Certificate of Analysis covering viability, potency, purity, and sterility.
Deliberate design for predictable cell potency, product safety, and ease of clinical use.
Our work is grounded in a commitment to patient safety and responsible clinical impact. In a category often marked by variability and eneven standards, TAVRA takes an intentionally conservative approach that is anchored in biological rationale, manufacturing rigor, and transparency.
How TAVRA Supports Clinical Care
Built on the most studied mesenchymal stem cells, supported by FDA-approved therapeutic history.
TAVRA supplies stem cell-based biologic products to licensed physicians for localized clinical use. Our focus is on supporting orthopedic, sports medicine, and pain focused clinical practices where regenerative therapies may meaningfully support tissue health, function, and recovery.


TAVRA Biosciences products are not approved by the U.S. Food and Drug Administration. Products are supplied exclusively to licensed physicians for localized clinical use within applicable regulatory frameworks. Information on this website is provided for educational purposes only.